Showing Spotlights 25 - 32 of 137 in category All (newest first):
 The Action Plan, presented by the EU Commission in 2004, envisioned integrating "the social dimension into a responsible technology development" and strengthening efforts related to "health, safety, environmental aspects and consumer protection". This encompassed (1) the systematic study of safety-relevant aspects at the earliest possible date, (2) integrating health- and environment-relevant aspect in research and development, (3) conducting targeted studies on toxicology and ecotoxicology and, finally, (4) adapting risk assessment approaches to nano-specific aspects in all phases of product life-cycles. The primary goal was to improve the competitiveness of European industry. The draft presented in mid-2011 for the planned research priorities2 continues this strategic focus. This article describes a selection of 22 current projects dealing with safety research as related to nanotechnology.
The Action Plan, presented by the EU Commission in 2004, envisioned integrating "the social dimension into a responsible technology development" and strengthening efforts related to "health, safety, environmental aspects and consumer protection". This encompassed (1) the systematic study of safety-relevant aspects at the earliest possible date, (2) integrating health- and environment-relevant aspect in research and development, (3) conducting targeted studies on toxicology and ecotoxicology and, finally, (4) adapting risk assessment approaches to nano-specific aspects in all phases of product life-cycles. The primary goal was to improve the competitiveness of European industry. The draft presented in mid-2011 for the planned research priorities2 continues this strategic focus. This article describes a selection of 22 current projects dealing with safety research as related to nanotechnology.
Jun 18th, 2012
 A substance might potentially be harmful or even toxic for a biological system, provided that the quantity or the concentration (the "dose") is high enough. This principle forms the basis for health standards which determine the maximum permissible concentration of contaminations, for example in food, water or in the environment. Dose calculation is of high relevance for risk assessment as well as for regulations, for instance to determine the maximum allowable concentration of chemicals and particles or to determine other limits which do not cause health problems. In the case of nanomaterials, especially for nanoparticles there is to date, no limit regulation or any other regulation referring to dose, because the definition of dose for nanoparticles does not exist. The wherefores will be explained in this dossier.
A substance might potentially be harmful or even toxic for a biological system, provided that the quantity or the concentration (the "dose") is high enough. This principle forms the basis for health standards which determine the maximum permissible concentration of contaminations, for example in food, water or in the environment. Dose calculation is of high relevance for risk assessment as well as for regulations, for instance to determine the maximum allowable concentration of chemicals and particles or to determine other limits which do not cause health problems. In the case of nanomaterials, especially for nanoparticles there is to date, no limit regulation or any other regulation referring to dose, because the definition of dose for nanoparticles does not exist. The wherefores will be explained in this dossier.
May 24th, 2012
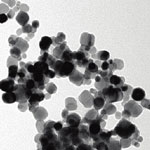 Several studies in the literature have highlighted that as nanomaterials "age" they can undergo oxidation; sintering (coalescence); surface ligand displacement; smaller nanoparticle formation; and surface carbonate formation. Nevertheless, no studies are available on how these changes affect the physicochemical properties of the nanomaterials. The aging of nanomaterials is expected to be rapid even under ambient environmental conditions. With the consequence that pristine, as synthesized materials - which are commonly used in nanotechnology-relevant environmental health and safety (EHS) studies - are never really encountered under natural environmental conditions. Which means that researchers who investigate the applications and implications of nanomaterials need to have a clear understanding of the aging process of these materials and need to take its effects into consideration.
Several studies in the literature have highlighted that as nanomaterials "age" they can undergo oxidation; sintering (coalescence); surface ligand displacement; smaller nanoparticle formation; and surface carbonate formation. Nevertheless, no studies are available on how these changes affect the physicochemical properties of the nanomaterials. The aging of nanomaterials is expected to be rapid even under ambient environmental conditions. With the consequence that pristine, as synthesized materials - which are commonly used in nanotechnology-relevant environmental health and safety (EHS) studies - are never really encountered under natural environmental conditions. Which means that researchers who investigate the applications and implications of nanomaterials need to have a clear understanding of the aging process of these materials and need to take its effects into consideration.
Mar 8th, 2012
 A group of experts from the chemical industry and various research laboratories in Germany have published a report on the current status of risk research on nanotechnology materials and applications. The report - 10 Years of Research: Risk Assessment, Human and Environmental Toxicology of Nanomaterials - provides an overview of the current state of risk assessment and toxicological research into nanomaterials. It also lists and summarizes the national and European projects on toxicology on various nanomaterials. In their report, the working group "Responsible Production and Use of Nanomaterials" has drawn up a list of topics and priorities which need to be addressed; activities and projects which have already been carried out; are currently on-going; or are still at the planning stage. The main focus of our considerations is on Germany, with a wider outlook on papers and results at European level.
A group of experts from the chemical industry and various research laboratories in Germany have published a report on the current status of risk research on nanotechnology materials and applications. The report - 10 Years of Research: Risk Assessment, Human and Environmental Toxicology of Nanomaterials - provides an overview of the current state of risk assessment and toxicological research into nanomaterials. It also lists and summarizes the national and European projects on toxicology on various nanomaterials. In their report, the working group "Responsible Production and Use of Nanomaterials" has drawn up a list of topics and priorities which need to be addressed; activities and projects which have already been carried out; are currently on-going; or are still at the planning stage. The main focus of our considerations is on Germany, with a wider outlook on papers and results at European level.
Feb 28th, 2012
 Following up on our recent Nanowerk Spotlight on nanofoods, new research shows that consumers could be exposed to nanoparticles in food by a much larger degree than has been expected so far. For a modern consumer it is hard to avoid titanium dioxide (TiO2) - a widely used additive in food, personal care and other household products. Approximately 7 million tons of bulk TiO2 are produced annually and used as white pigment in order to provide whiteness and opacity to foods and other products. Many applications of titanium dioxide would benefit from smaller primary particle sizes, and we can expect the percentage of TiO2 that is produced in or near the nano range to increase.
Following up on our recent Nanowerk Spotlight on nanofoods, new research shows that consumers could be exposed to nanoparticles in food by a much larger degree than has been expected so far. For a modern consumer it is hard to avoid titanium dioxide (TiO2) - a widely used additive in food, personal care and other household products. Approximately 7 million tons of bulk TiO2 are produced annually and used as white pigment in order to provide whiteness and opacity to foods and other products. Many applications of titanium dioxide would benefit from smaller primary particle sizes, and we can expect the percentage of TiO2 that is produced in or near the nano range to increase.
Feb 15th, 2012
 In a project funded by the Danish Environmental Protection Agency, researchers have initiated the development of a screening tool called NanoRiskCat for the evaluation of exposure and hazard of nanomaterials contained in products for professional and private use. The project's aim was to identify, categorize and rank the possible exposure and hazards associated with a nanomaterial in a product. NanoRiskCat is using a stepwise approach based on existing data on the conventional form of the chemical as well as the data that may exist on the nanoform. However, the tool still needs to be further validated and tested on a series of various nano products in order to adjust and optimize the concept and thereby to achieve a screening tool as informative and practical as possible.
In a project funded by the Danish Environmental Protection Agency, researchers have initiated the development of a screening tool called NanoRiskCat for the evaluation of exposure and hazard of nanomaterials contained in products for professional and private use. The project's aim was to identify, categorize and rank the possible exposure and hazards associated with a nanomaterial in a product. NanoRiskCat is using a stepwise approach based on existing data on the conventional form of the chemical as well as the data that may exist on the nanoform. However, the tool still needs to be further validated and tested on a series of various nano products in order to adjust and optimize the concept and thereby to achieve a screening tool as informative and practical as possible.
Jan 25th, 2012
 Early detection of food borne pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. Conventional techniques to detect E. coli and other pathogenic bacteria are time-consuming, labor-intensive, and inadequate as they lack the ability to detect bacteria in real time. Thus, there is an urgent need for alternative platforms for the rapid, sensitive, reliable and simple isolation and detection pathogens. Taking a novel approach to isolating pathogenic bacteria from complex clinical, environmental and food samples, researchers have developed a nanomotor strategy that involves the movement of lectin-functionalized microengines. Receptor-functionalized nanoswimmers offer direct and rapid target isolation from raw biological samples without preparatory and washing steps.
Early detection of food borne pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. Conventional techniques to detect E. coli and other pathogenic bacteria are time-consuming, labor-intensive, and inadequate as they lack the ability to detect bacteria in real time. Thus, there is an urgent need for alternative platforms for the rapid, sensitive, reliable and simple isolation and detection pathogens. Taking a novel approach to isolating pathogenic bacteria from complex clinical, environmental and food samples, researchers have developed a nanomotor strategy that involves the movement of lectin-functionalized microengines. Receptor-functionalized nanoswimmers offer direct and rapid target isolation from raw biological samples without preparatory and washing steps.
Dec 8th, 2011
 Naturally occurring nanomaterials can be found everywhere in nature and only with recent advances in instrumentation and metrology equipment are researchers beginning to locate, isolate, characterize and classify the vast range of their structural and chemical varieties. Scientists are beginning to recognize that all sources of nanomaterials are important in evaluating the possible impact of nanoscale materials on human health and the environment; however, perhaps the greatest benefit to studying these materials will be in their ability to inform researchers about the manner in which nano-sized materials have been a part of our environment from the beginning.
Naturally occurring nanomaterials can be found everywhere in nature and only with recent advances in instrumentation and metrology equipment are researchers beginning to locate, isolate, characterize and classify the vast range of their structural and chemical varieties. Scientists are beginning to recognize that all sources of nanomaterials are important in evaluating the possible impact of nanoscale materials on human health and the environment; however, perhaps the greatest benefit to studying these materials will be in their ability to inform researchers about the manner in which nano-sized materials have been a part of our environment from the beginning.
Nov 24th, 2011
 The Action Plan, presented by the EU Commission in 2004, envisioned integrating "the social dimension into a responsible technology development" and strengthening efforts related to "health, safety, environmental aspects and consumer protection". This encompassed (1) the systematic study of safety-relevant aspects at the earliest possible date, (2) integrating health- and environment-relevant aspect in research and development, (3) conducting targeted studies on toxicology and ecotoxicology and, finally, (4) adapting risk assessment approaches to nano-specific aspects in all phases of product life-cycles. The primary goal was to improve the competitiveness of European industry. The draft presented in mid-2011 for the planned research priorities2 continues this strategic focus. This article describes a selection of 22 current projects dealing with safety research as related to nanotechnology.
The Action Plan, presented by the EU Commission in 2004, envisioned integrating "the social dimension into a responsible technology development" and strengthening efforts related to "health, safety, environmental aspects and consumer protection". This encompassed (1) the systematic study of safety-relevant aspects at the earliest possible date, (2) integrating health- and environment-relevant aspect in research and development, (3) conducting targeted studies on toxicology and ecotoxicology and, finally, (4) adapting risk assessment approaches to nano-specific aspects in all phases of product life-cycles. The primary goal was to improve the competitiveness of European industry. The draft presented in mid-2011 for the planned research priorities2 continues this strategic focus. This article describes a selection of 22 current projects dealing with safety research as related to nanotechnology.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





