Showing Spotlights 9 - 16 of 77 in category All (newest first):
 Researchers estimate that global PV module waste, 90% of which are made from crystalline silicon, will reach 1.7-1.8 million tonnes by 2030 and 60-78 million tonnes by 2050, which will likely reach the same order of magnitude as global electronic waste. A novel upcycling strategy adds value to this otherwise economically low-value waste silicon. With proper chemical doping, it allows silicon waste to be turned into high-performance thermoelectrics for heat energy harvesting.
Researchers estimate that global PV module waste, 90% of which are made from crystalline silicon, will reach 1.7-1.8 million tonnes by 2030 and 60-78 million tonnes by 2050, which will likely reach the same order of magnitude as global electronic waste. A novel upcycling strategy adds value to this otherwise economically low-value waste silicon. With proper chemical doping, it allows silicon waste to be turned into high-performance thermoelectrics for heat energy harvesting.
May 16th, 2022
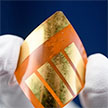 Organic and perovskite solar cells are very promising energy harvester for indoor Internet-of-Things (IoT) applications as the requirements that solar cells should satisfy to power IoT devices are quite different to the ones usually deemed necessary for application in outdoor-placed solar panels. The size of the single solar cell used in IoT applications is much smaller, and in combination with the lower power input available in low-light indoor settings as well as the emission spectra of light sources other than the sun, renders the need for high conversion efficiency paramount.
Organic and perovskite solar cells are very promising energy harvester for indoor Internet-of-Things (IoT) applications as the requirements that solar cells should satisfy to power IoT devices are quite different to the ones usually deemed necessary for application in outdoor-placed solar panels. The size of the single solar cell used in IoT applications is much smaller, and in combination with the lower power input available in low-light indoor settings as well as the emission spectra of light sources other than the sun, renders the need for high conversion efficiency paramount.
Apr 28th, 2022
 Perovskite semiconductor solar cells are a very exciting photovoltaic technology possessing similar efficiencies to silicon but cast or printed in thin films via liquid inks. Researchers have developed a new method that uses a simple sheet of paper to deposit the perovskite films that does without any expensive equipment. The trick to achieve high performance with this remarkably cheap method is to soak the paper applicator in anti-solvent which almost doubles efficiencies compared to when using it dry, reaching 11% on flexible plastic substrates.
Perovskite semiconductor solar cells are a very exciting photovoltaic technology possessing similar efficiencies to silicon but cast or printed in thin films via liquid inks. Researchers have developed a new method that uses a simple sheet of paper to deposit the perovskite films that does without any expensive equipment. The trick to achieve high performance with this remarkably cheap method is to soak the paper applicator in anti-solvent which almost doubles efficiencies compared to when using it dry, reaching 11% on flexible plastic substrates.
Feb 15th, 2022
 Scientists have demonstrated a technique for producing perovskite photovoltaic materials on an industrial scale, which will reduce the cost and improve the performance of mass-produced perovskite solar cells. The technique, which uses spin coating by designing a co-solvent dilution strategy, is low-cost, simple, energy-efficient, and should pave the way for creating perovskite solar cells. This work demonstrates that these existing technologies could be used to create perovskite solar cells.
Scientists have demonstrated a technique for producing perovskite photovoltaic materials on an industrial scale, which will reduce the cost and improve the performance of mass-produced perovskite solar cells. The technique, which uses spin coating by designing a co-solvent dilution strategy, is low-cost, simple, energy-efficient, and should pave the way for creating perovskite solar cells. This work demonstrates that these existing technologies could be used to create perovskite solar cells.
Jan 31st, 2022
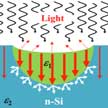 New research shows the formation of large-area R8-Si within the top n-layer of the p-n junction Si solar cell using spherical nanoindentation, and experimentally measured optical properties of R8-Si to demonstrate ten times enhancement in its photocurrent density. After theoretical studies in 2008 indicated that rhombohedral phase (R8) of silicon, which is stable at ambient temperature, has interesting optoelectronic properties suitable for a solar absorber application, this is the first demonstration in experimental studies.
New research shows the formation of large-area R8-Si within the top n-layer of the p-n junction Si solar cell using spherical nanoindentation, and experimentally measured optical properties of R8-Si to demonstrate ten times enhancement in its photocurrent density. After theoretical studies in 2008 indicated that rhombohedral phase (R8) of silicon, which is stable at ambient temperature, has interesting optoelectronic properties suitable for a solar absorber application, this is the first demonstration in experimental studies.
Apr 17th, 2020
 New work provides insight into the control over phase and ordering during halide perovskite epitaxial growth and expands the selection of photoactive materials for growing epitaxial halide perovskites that can be exploited in high-performance electronic applications. Hybrid organic-inorganic halide perovskite has attracted tremendous attention as an exceptional new class of semiconductors for solar harvesting, light emission, lasing and thin-film electronics. However, the toxicity of lead devices and lead manufacturing combined with the instability of organic components have been two key barriers to widespread application. Tin-based inorganic halide perovskites have been considered promising substitutes for their lead analogues.
New work provides insight into the control over phase and ordering during halide perovskite epitaxial growth and expands the selection of photoactive materials for growing epitaxial halide perovskites that can be exploited in high-performance electronic applications. Hybrid organic-inorganic halide perovskite has attracted tremendous attention as an exceptional new class of semiconductors for solar harvesting, light emission, lasing and thin-film electronics. However, the toxicity of lead devices and lead manufacturing combined with the instability of organic components have been two key barriers to widespread application. Tin-based inorganic halide perovskites have been considered promising substitutes for their lead analogues.
Nov 7th, 2019
 Due to their fascinating optoelectronic properties, halide perovskites have attracted tremendous research interest as promising materials for photovoltaics, photodetectors, LEDs, and lasers. Researchers report report a strategy to synthesize perovskite nanocrystals using diblock copolymer micelles as a growth template. This work constitutes a new approach for synthesizing perovskite nanocrystals of controllable size and composition with vastly improved resistance to halide ion migration and environmentally induced degradation.
Due to their fascinating optoelectronic properties, halide perovskites have attracted tremendous research interest as promising materials for photovoltaics, photodetectors, LEDs, and lasers. Researchers report report a strategy to synthesize perovskite nanocrystals using diblock copolymer micelles as a growth template. This work constitutes a new approach for synthesizing perovskite nanocrystals of controllable size and composition with vastly improved resistance to halide ion migration and environmentally induced degradation.
Aug 7th, 2019
 Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
Feb 7th, 2019
 Researchers estimate that global PV module waste, 90% of which are made from crystalline silicon, will reach 1.7-1.8 million tonnes by 2030 and 60-78 million tonnes by 2050, which will likely reach the same order of magnitude as global electronic waste. A novel upcycling strategy adds value to this otherwise economically low-value waste silicon. With proper chemical doping, it allows silicon waste to be turned into high-performance thermoelectrics for heat energy harvesting.
Researchers estimate that global PV module waste, 90% of which are made from crystalline silicon, will reach 1.7-1.8 million tonnes by 2030 and 60-78 million tonnes by 2050, which will likely reach the same order of magnitude as global electronic waste. A novel upcycling strategy adds value to this otherwise economically low-value waste silicon. With proper chemical doping, it allows silicon waste to be turned into high-performance thermoelectrics for heat energy harvesting.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





